Angiopoietin-like protein 8 orchestrates macrophage glycogen metabolism and polarization via the JNK signaling pathway in cytokine storm syndrome
血管生成素样蛋白8通过JNK信号通路调控细胞因子风暴综合征中的巨噬细胞糖原代谢和极化
| 期刊: | Cell and Bioscience | 影响因子: | 6.100 |
| 时间: | 2025 | 起止号: | 2025 Oct 15;15(1):140. |
| doi: | 10.1186/s13578-025-01487-7 | 研究方向: | 代谢 |
文献解析
1. 领域背景与文献
文献英文标题:Angiopoietin-like 8 promotes macrophage M1 polarization via JNK-mediated glycogen metabolism to drive cytokine storm syndrome;发表期刊:BMC Complementary Medicine and Therapies;影响因子:3.9(2023年);研究领域:细胞因子风暴综合征与巨噬细胞极化调控
细胞因子风暴综合征(CSS)是急性感染、自身免疫疾病等病理过程中出现的严重全身炎症反应,可导致多器官损伤甚至死亡,在COVID-19大流行期间其死亡率可达25%,是临床亟待解决的难题。领域共识:当前CSS临床治疗以多学科综合干预为主,包括抗菌抗炎、器官支持等手段,但存在抗生素与激素滥用引发的并发症、呼吸干预损伤等问题,亟需更安全有效的治疗策略。巨噬细胞的M1/M2极化失衡是CSS的核心病理机制,M1型巨噬细胞过度活化会大量释放促炎细胞因子,引发组织损伤;而M2型巨噬细胞则发挥抗炎修复作用,维持免疫稳态。肝脏作为重要的免疫应答器官,分泌的肝细胞因子可调控巨噬细胞活化,如成纤维细胞生长因子21(FGF-21)可通过抑制M1极化发挥保护作用,但重组FGF-21半衰期短的局限性限制了其临床应用。因此,寻找新型肝细胞因子调控巨噬细胞极化,成为CSS治疗领域的研究空白与核心需求。本研究聚焦血管生成素样蛋白8(Angptl8),旨在明确其在CSS中的作用及调控机制,为CSS治疗提供新靶点。
2. 文献综述解析
作者从CSS临床治疗困境、巨噬细胞极化的核心作用、肝脏肝细胞因子的调控潜力三个维度对现有研究进行分类综述。现有研究已明确CSS的高死亡率与巨噬细胞过度活化直接相关,临床多学科干预虽能覆盖CSS的多病理环节,但存在诸多并发症;肝脏分泌的肝细胞因子如FGF-21可通过调控巨噬细胞极化发挥保护作用,但存在药效持续时间短的局限性;同时,Angptl8作为代谢调控因子,其在炎症及CSS中的作用尚未被系统解析。现有技术方法中,细胞与动物模型可有效解析巨噬细胞极化机制,但缺乏针对CSS特异性调控因子的深入研究,且已有的治疗靶点存在明显的应用限制。
本研究的创新价值在于首次揭示了Angptl8在炎症条件下可作为肝细胞因子及巨噬细胞来源的分泌蛋白,通过JNK介导的糖原代谢通路促进巨噬细胞M1极化、抑制M2极化,从而驱动CSS进展;首次验证了Angptl8中和抗体可有效缓解CSS症状,降低死亡率,为CSS治疗提供了全新的靶点与候选药物,填补了Angptl8在CSS领域研究的空白,突破了现有治疗手段的局限性。
3. 研究思路总结与详细解析
本研究的核心目标是明确Angptl8在LPS诱导的CSS中的作用及调控机制,核心科学问题为Angptl8如何通过调控巨噬细胞极化影响CSS进展,技术路线遵循“表达特征分析→体内功能验证→细胞机制解析→通路筛选与验证→治疗效果验证”的闭环逻辑,从体内到体外系统解析Angptl8的功能与机制。
3.1 CSS模型中Angptl8表达特征分析
实验目的是明确LPS诱导的CSS中Angptl8的表达变化及细胞来源。研究构建LPS诱导的小鼠CSS模型,采用25mg/kg LPS腹腔注射处理小鼠,通过ELISA、qRT-PCR、Western blot等方法检测血清、肝脏、骨髓中Angptl8的表达水平;同时分离小鼠原代肝细胞(PHs)和骨髓来源巨噬细胞(BMDMs),经LPS处理后检测Angptl8的表达及分泌情况。
结果显示,LPS处理后小鼠血清Angptl8水平显著升高;肝脏中Angptl8 mRNA和蛋白分别上调12.1倍(n=6,P<0.01)和4.2倍(n=3,P<0.01);PHs和BMDMs中LPS以剂量和时间依赖方式诱导Angptl8的mRNA和蛋白表达,且细胞上清中Angptl8分泌量显著增加;骨髓中Angptl8 mRNA上调14.4倍。这些结果表明Angptl8在CSS中显著上调,其来源包括肝脏和巨噬细胞。

实验所用关键产品:ABclonal的巨噬细胞集落刺激因子(M-CSF)、BD FACSFortessa流式细胞仪、Sigma的胶原酶IV、Cloud-Clone的重组小鼠His-Angptl8蛋白等。
3.2 Angptl8敲除对CSS的体内保护作用验证
实验目的是验证Angptl8缺失是否能缓解LPS诱导的CSS症状。研究将Angptl8敲除(Angptl8-/-)小鼠和野生型(WT)小鼠用25mg/kg LPS处理,观察72小时生存率;通过ELISA检测血清中TNF-α、IL-6、IFN-γ等促炎细胞因子水平;采用qRT-PCR检测肝脏、脾脏、骨髓中促炎细胞因子的mRNA表达;通过H&E染色、TUNEL染色观察肝脾组织损伤情况;检测血清中ALT、AST等肝酶水平评估肝功能。
结果显示,Angptl8-/-小鼠的生存率从WT小鼠的0提升至60%(n=10,P<0.01);血清中TNF-α、IL-6、IFN-γ水平显著降低;肝脏、脾脏、骨髓中Tnf-α、Il-6、Il-1β、Inos的mRNA表达均显著下调;H&E染色显示Angptl8-/-小鼠肝小叶结构损伤减轻,桥接坏死和炎症细胞浸润减少;TUNEL染色显示肝细胞凋亡数量显著减少;血清ALT、AST水平也相应降低。这些结果表明Angptl8缺失可显著缓解CSS症状,降低小鼠死亡率。

文献未提及具体实验产品,领域常规使用ELISA试剂盒、组织学染色试剂盒等。
3.3 Angptl8对巨噬细胞极化的调控作用解析
实验目的是明确Angptl8对巨噬细胞M1/M2极化的直接调控作用。研究分离WT和Angptl8-/-小鼠的BMDMs,经LPS诱导M1极化后,通过流式细胞术检测CD86+细胞比例,qRT-PCR和Western blot检测促炎细胞因子的表达;用重组Angptl8处理BMDMs,检测M1极化标志物的变化;分离WT和Angptl8-/-小鼠的库普弗细胞,通过免疫荧光检测CD86(M1标志物)和CD206(M2标志物)的表达;同时用IL-4诱导BMDMs M2极化,检测Angptl8缺失或重组Angptl8处理对CD206+细胞比例及M2标志物表达的影响。
结果显示,Angptl8-/-小鼠BMDMs中LPS诱导的CD86+细胞比例显著降低,促炎细胞因子的mRNA和蛋白表达均下调;重组Angptl8以剂量依赖方式增加CD86+细胞比例,上调促炎细胞因子表达;Angptl8-/-小鼠库普弗细胞中LPS诱导的CD86表达降低,CD206表达升高;Angptl8缺失可增强IL-4诱导的M2极化,而重组Angptl8则抑制M2极化。这些结果表明Angptl8是促进巨噬细胞M1极化、抑制M2极化的关键调控因子。

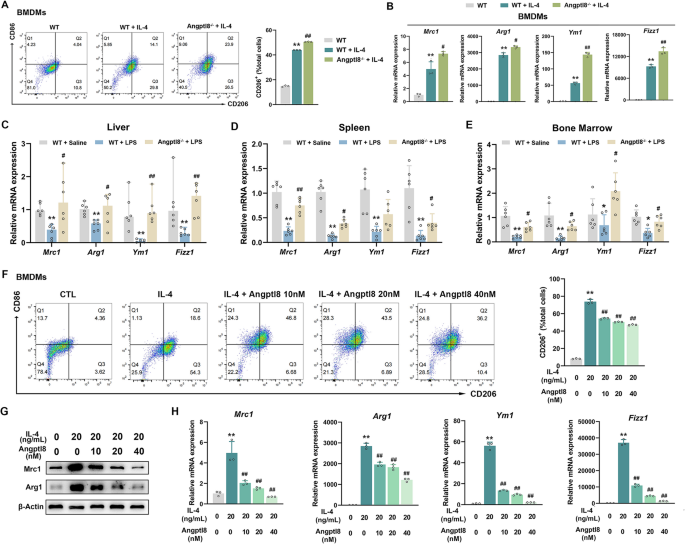
实验所用关键产品:Biolegend的CD86、CD206抗体,Invitrogen的F4/80抗体,Peprotech的IL-4等。
3.4 Angptl8调控巨噬细胞极化的下游通路筛选与验证
实验目的是解析Angptl8调控巨噬细胞极化的分子机制。研究对LPS处理的WT和Angptl8-/-小鼠骨髓及BMDMs进行转录组测序,通过GO和GSEA分析富集通路;采用PAS染色和透射电镜(TEM)检测BMDMs中的糖原水平;检测糖原代谢相关酶(Pgm1、Ugp2、Gys1、Pygm)的mRNA表达;用糖原磷酸化酶抑制剂GPI处理BMDMs,验证糖原代谢在Angptl8调控M1极化中的作用;通过KEGG分析筛选JNK通路,采用Western blot检测JNK磷酸化水平;用JNK抑制剂SP600125处理,验证JNK在Angptl8调控糖原代谢和M1极化中的作用。
结果显示,转录组分析显示糖原代谢和炎症反应通路显著富集;Angptl8-/-小鼠BMDMs中LPS诱导的糖原积累减少,糖原代谢相关酶的mRNA表达下调;重组Angptl8可增加BMDMs糖原水平,上调糖原分解酶Pygm的表达;GPI处理可抑制Angptl8诱导的M1极化,恢复Angptl8对M2极化的抑制作用;KEGG分析显示JNK通路富集,重组Angptl8可促进JNK磷酸化;SP600125处理可抑制Angptl8诱导的糖原积累和M1极化,恢复M2极化。这些结果表明Angptl8通过JNK介导的糖原代谢通路调控巨噬细胞极化。

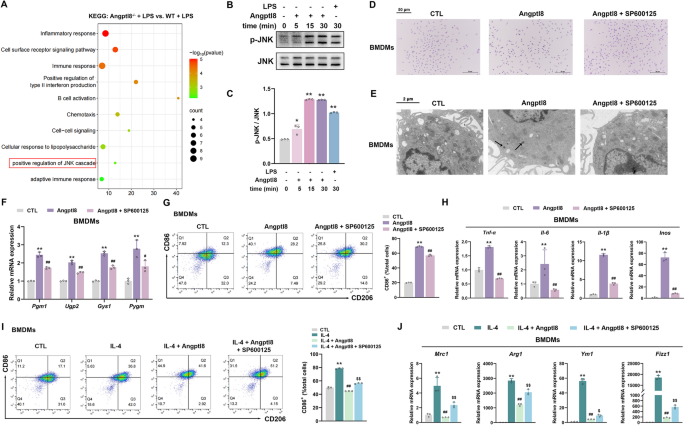
实验所用关键产品:Beyotime的PAS试剂盒、Hitachi HT7800透射电镜、GraphPad Prism 9.5统计软件等。
3.5 Angptl8中和抗体的CSS治疗效果验证
实验目的是验证Angptl8中和抗体在体内对CSS的治疗作用及安全性。研究在LPS处理前2小时给小鼠静脉注射Angptl8中和抗体(10μg/25g、50μg/25g),观察小鼠72小时生存率;通过ELISA检测血清促炎细胞因子水平;采用qRT-PCR检测肝脏、脾脏、骨髓中促炎细胞因子的mRNA表达;通过H&E染色、TUNEL染色观察肝脾组织损伤情况;检测血清中肝、肾、心功能标志物,评估多器官损伤情况。
结果显示,中和抗体处理后小鼠生存率提升至60%(n=10,P<0.01);血清中TNF-α、IL-6、IFN-γ水平显著降低;肝脏、脾脏、骨髓中促炎细胞因子的mRNA表达均显著下调;TUNEL和H&E染色显示肝脾组织损伤减轻;血清中ALT、AST、肌酐、尿素氮(BUN)、心房钠尿肽(ANP)、脑钠肽(BNP)水平降低,肾、心、肺组织学损伤也明显减轻。这些结果表明Angptl8中和抗体可有效缓解CSS症状,且无明显的体内毒性。
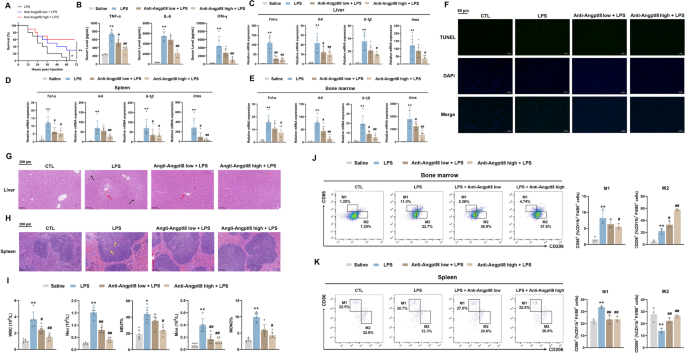
实验所用关键产品:Bioworld的Angptl8中和抗体等。
4. Biomarker研究及发现成果
Biomarker定位
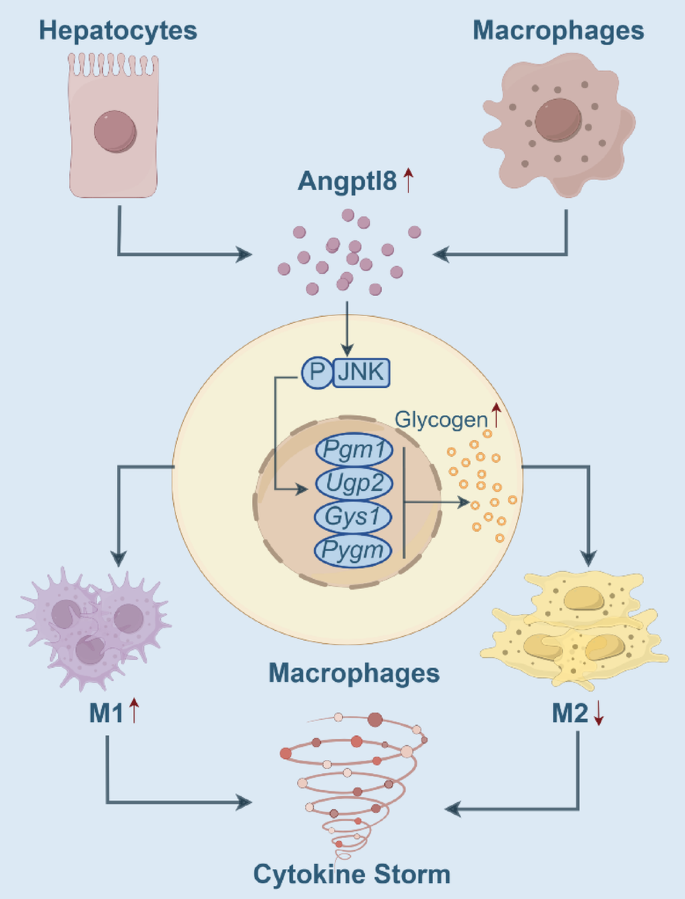
本研究中Angptl8属于功能性Biomarker,其筛选与验证逻辑为:首先在LPS诱导的CSS模型中发现Angptl8表达显著上调;随后通过体内敲除实验验证其对CSS的调控作用;再通过细胞实验明确其对巨噬细胞极化的直接调控;最后通过转录组测序与功能实验验证其调控通路,并验证中和抗体的治疗效果,形成完整的“表达-功能-机制-治疗”验证链条。
研究过程详述
Angptl8的来源包括肝脏肝细胞和炎症状态下的巨噬细胞,研究通过ELISA定量检测血清Angptl8水平,qRT-PCR和Western blot验证组织与细胞中的表达,流式细胞术检测其对巨噬细胞极化的调控作用,转录组测序筛选下游通路。特异性方面,Angptl8在CSS模型中特异性上调,且其表达变化与巨噬细胞极化程度及CSS严重程度正相关;敏感性方面,LPS处理后短时间内即可检测到Angptl8的表达上调,可作为CSS早期的响应标志物。
核心成果提炼
Angptl8作为CSS的功能性Biomarker,其核心功能是通过JNK介导的糖原代谢通路促进巨噬细胞M1极化,驱动CSS进展,其缺失或中和可显著降低CSS小鼠死亡率(生存率从0提升至60%,n=10,P<0.01),下调促炎细胞因子水平,减轻多器官损伤。本研究首次在CSS中发现Angptl8与巨噬细胞糖原代谢的调控关系,其创新性在于明确了Angptl8从代谢调控因子向免疫调控因子的功能转换,为CSS治疗提供了全新的靶点,Angptl8中和抗体具有良好的临床应用潜力,尤其适用于病毒感染及免疫治疗相关的CSS患者。
特别声明
1、本页面内容包含部分的内容是基于公开信息的合理引用;引用内容仅为补充信息,不代表本站立场。
2、若认为本页面引用内容涉及侵权,请及时与本站联系,我们将第一时间处理。
3、其他媒体/个人如需使用本页面原创内容,需注明“来源:[生知库]”并获得授权;使用引用内容的,需自行联系原作者获得许可。
4、投稿及合作请联系:info@biocloudy.com。